Medical device testing machine
Reliable verification is a core part of maintaining medical equipment performance, supporting compliance workflows, and reducing risk in clinical and laboratory environments. From biomedical analyzers used during preventive maintenance to imaging phantoms for system validation and training, the right tools help technicians check whether a device behaves as expected before it returns to service.
Medical device testing machine solutions in this category cover a broad range of practical needs. That includes performance testing for defibrillation-related equipment, simulation-based checks, and phantom-based evaluation for imaging applications such as X-ray CT, ultrasound, and MRI.
Where these testing systems fit in practice
Biomedical and healthcare engineering teams use test equipment to verify device output, compare measured values against expected ranges, and document results for service records. In many cases, the objective is not only fault finding, but also routine quality control after repair, calibration, transport, or scheduled maintenance.
This category brings together instruments used across different workflows. Some devices are intended for direct analyzer-based checks, while others are designed as phantoms that emulate tissue, anatomical structures, or imaging conditions so that CT, ultrasound, and MRI systems can be evaluated in a repeatable way.
Key product groups in this category
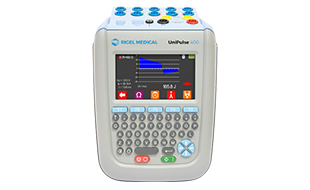
One important group is defibrillation and pacing analysis. A representative example is the Fluke (Biomedical) Impulse 7000DP Defibrillator/Transcutaneous Pacer Analyzer, which is designed for checking critical output parameters in defibrillator and transcutaneous pacer applications. Tools in this area are relevant when service teams need to verify delivered pulse characteristics and confirm that emergency equipment remains within acceptable performance limits.
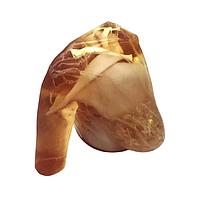
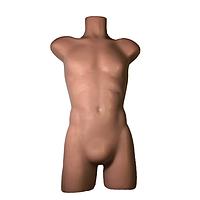
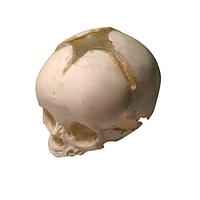
Another major group is imaging phantoms. Products from True Phantom Solutions illustrate how phantom-based testing supports modality validation, research, training, and protocol development. Depending on the application, users may need a Doppler phantom, an anatomical rat model, a newborn head, torso, arm, or a larger animal phantom for cross-modality studies.
Examples of imaging phantom applications
Phantoms are especially useful when teams need a controlled target instead of relying on patient-dependent variables. The True Phantom Solutions DP-C01 Doppler Phantom For X-Ray CT, US, MRI is an example of a model intended for multi-modality work, helping users evaluate image behavior and flow-related or tissue-equivalent responses under repeatable conditions.
For anatomical and preclinical-style workflows, examples in this category include the True Phantom Solutions RT-C01 Rat Phantom (Cylindrical) For X-Ray CT, MRI and the True Phantom Solutions RT-A02 Rat Phantom (Anatomical) For X-Ray CT, US. There are also neonatal-focused models such as the HD-N01, HD-N02, and HD-N03 newborn head variants, the AM-S01 newborn arm, and the AN-N01, AN-N02, and US-N02 newborn torso models. These products are relevant when users need tissue-mimicking structures for imaging evaluation, educational use, or method development.
How to choose the right medical device testing equipment
A practical starting point is the test objective. If the task is electrical or therapeutic output verification, an analyzer-oriented solution is typically appropriate. If the task involves image quality, anatomical visualization, tissue simulation, or modality comparison, a phantom-based solution is usually the better fit.
The next factor is modality coverage. Some products in this category are intended for X-ray CT and ultrasound, while others extend to MRI as well. Matching the phantom or analyzer to the equipment under test helps avoid unnecessary complexity and keeps the evaluation aligned with the real workflow.
Users should also consider sample form factor and use case. A compact tissue-equivalent insert or small anatomical model may be suitable for focused validation, while a larger and more detailed phantom may be more useful for protocol testing, research, or training. Where output values and safety-related behavior must be checked, teams may also compare their broader toolkit with instruments such as a multimeter for general electrical troubleshooting, although biomedical verification itself requires application-specific equipment.
Leading manufacturers represented in this range
Fluke (Biomedical) is widely recognized in biomedical test applications, especially where technicians need portable analyzers for maintenance and verification tasks. In this category, that expertise is represented by defibrillator and transcutaneous pacer analysis equipment suited to service environments that value repeatability, documentation, and routine field use.
Other notable brands in the broader category context include Raysafe, Rigel medical, and Sun Nuclear, alongside True Phantom Solutions. Each name is associated with different medical testing workflows, from biomedical verification to imaging-related quality assurance, allowing buyers to compare equipment according to actual application needs rather than brand alone.
Why repeatability matters in medical testing workflows
Medical equipment evaluation depends on consistent reference conditions. That is why analyzers and phantoms are so important: they provide a stable basis for checking performance over time, across different service events, and between multiple systems or locations. This is especially valuable in regulated environments where traceable maintenance records and standardized procedures are part of daily operations.
Repeatable testing also supports better decision-making. Instead of relying only on visual inspection or user feedback, service teams can measure outputs, observe imaging response, and compare trends from one test interval to the next. In that sense, these tools do more than identify failures; they help build a structured maintenance process.
Related measurement categories worth exploring
Some users working across biomedical engineering and technical maintenance may also need adjacent electrical and diagnostic tools. Depending on the job scope, related categories such as earth resistance/resistivity testers or a ground fault locator can be relevant for facility-level electrical investigations, even though they serve different purposes from dedicated medical test systems.
Keeping these categories separate is important: general electrical measurement tools support infrastructure and troubleshooting, while medical device testing equipment is used to evaluate clinical devices and imaging workflows in a more application-specific way.
Final considerations
Choosing the right solution in this category depends on what needs to be verified, how the test will be performed, and which modality or device type is involved. Analyzer-based tools are suited to output verification and service checks, while phantom-based products support imaging evaluation, training, and research under controlled conditions.
By focusing on application fit, modality compatibility, and repeatability, buyers can narrow this category more effectively and select equipment that supports safer maintenance routines and more consistent testing outcomes.
Get exclusive volume discounts, bulk pricing updates, and new product alerts delivered directly to your inbox.
By subscribing, you agree to our Terms of Service and Privacy Policy.
Direct access to our certified experts